 When an atom contains more or fewer electrons than protons, it has a negative or positive overall charge, and these atoms are known as ions.Īn atom is exceedingly small, much smaller than our imagination allows us to imagine. Like the layers of an anion, these electrons are organised in orbits around the nucleus of the atom. To have zero charge, an element's atom must have the same amount of protons as electrons. A single negative charge is carried by an electron. The total number of protons and neutrons in the nucleus is used to calculate an element's atomic weight. The number of protons or positive charges in the nucleus determines an element's atomic number. A proton is a particle with a single positive charge and a mass of one unit. A neutron is a neutral particle with a mass of one unit. The nucleus is made up of neutrons and protons, which are responsible for an atom's weight and positive charges. The atom is electrically neutral if the number of protons and electrons is equal. Protons have a positive electric charge, while electrons have a negative charge and neutrons don't have any. The nucleus contains more than 99.94% of an atom's mass. Only one type of hydrogen, the most prevalent, lacks neutrons. One or more protons and a number of neutrons make up the nucleus. This article will study atoms, molecules and ions, the difference between atom and molecule and molecular elements in detail.Ītoms Molecules and Ions Atoms and Molecules DefinitionĮvery atom is made up of a nucleus and one or more electrons attached to it. Atoms are extremely small, measuring about 100 picometers in diameter. Every solid, liquid, gas, and plasma is made up of neutral or ionised atoms. 1999‑2023 - All Rights Reserved.An atom is the smallest unit of matter that makes up a chemical element. Retrieved from Ĭopyright © Israel Science and Technology Directory. "Sortable list of elements of the Periodic Table". The story behind the discovery that elements are born in stars.Atomic Weights of the Elements (From IUPAC). 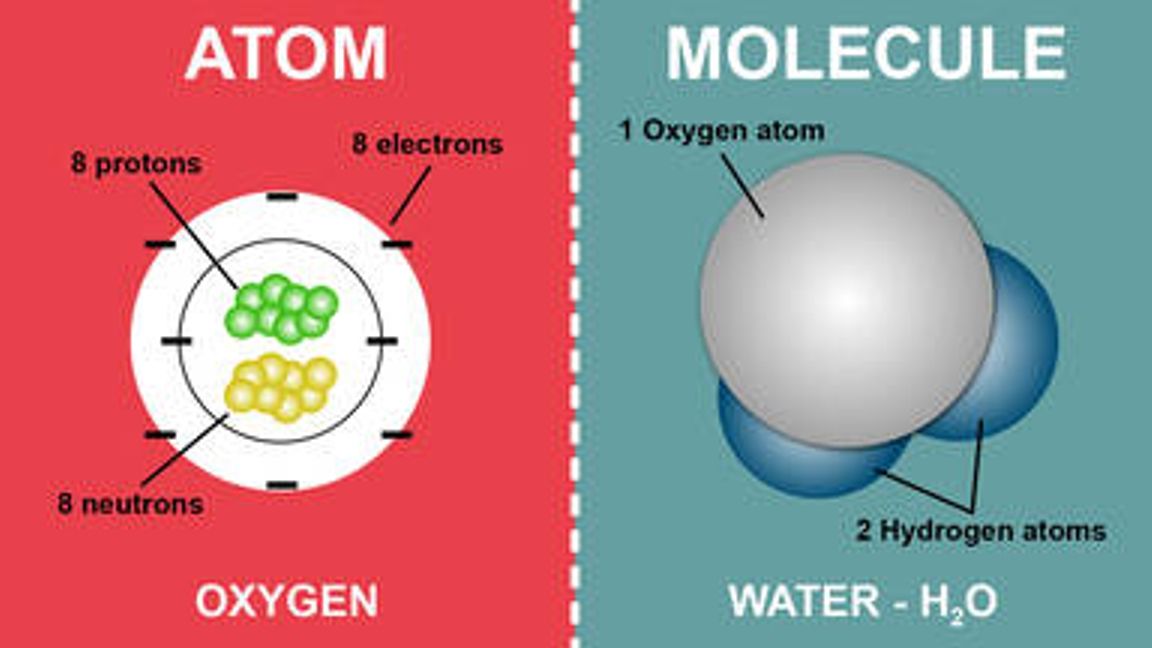
Multilingual Dictionary and Etymology of the Periodic Table Elements.Atomic Reference Data for Electronic Structure Calculations.List of Periodic Table Elements in Hebrew.Other resources related to the Periodic Table For these elements, the weight value represents the mass number of the longest-lived isotope of the element.Įlectron configuration: See next page for explanation of electron configuration of atoms. The elements marked with an asterisk have no stable nuclides. The values shown here are based on the IUPAC Commission determinations ( Pure Appl. For relative abundances of isotopes in nature, see reference on Atomic Weights and Isotopic Compositions.Ītomic weight: Atomic weight values represent weighted average of the masses of all naturally occurring isotopes of an element. The abundance of each isotope depends on the source of materials. For example, the two common isotopes of carbon, 12C and 13C, have 6 and 7 neutrons, respectively. Elements have more than one isotope with varying numbers of neutrons. The isotope of an element is defined by the sum of the number of protons and neutrons in its nucleus. Isotope: Atoms of the same element with the same atomic number, but a different number of neutrons. 
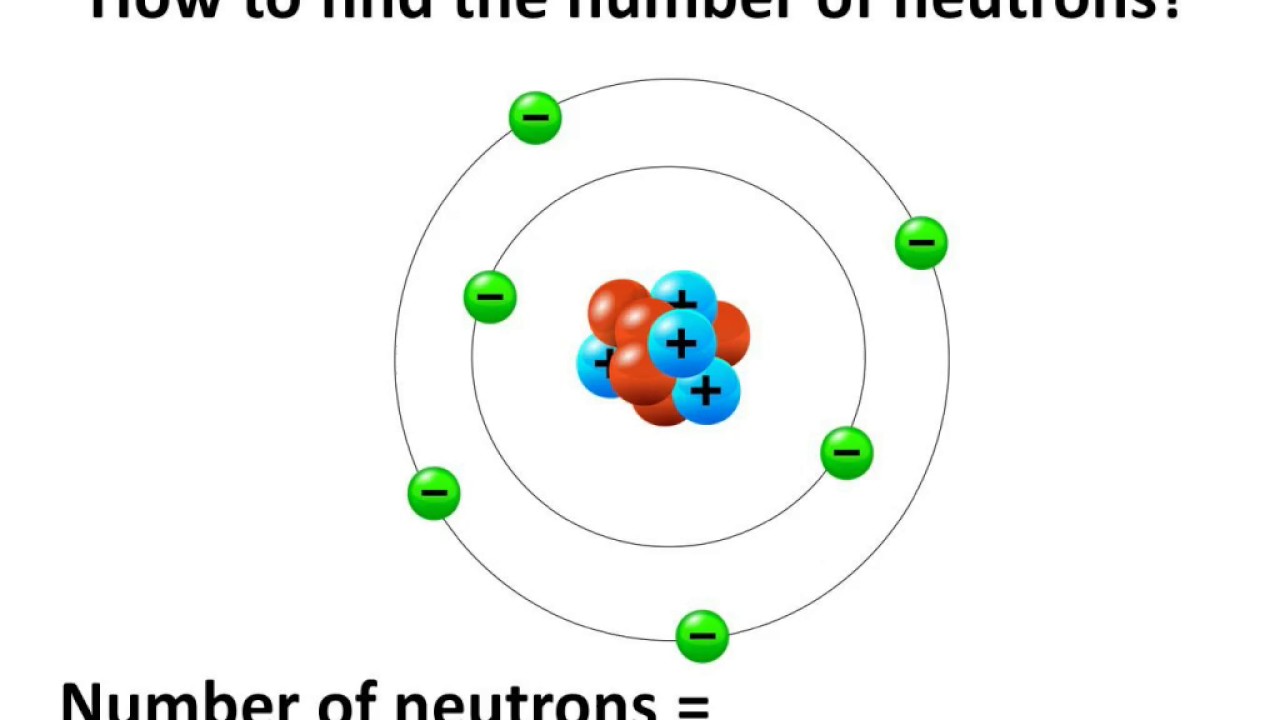
Thus, each proton and neutron has a mass of about 1 amu. This isotope of carbon has 6 protons and 6 neutrons. Atomic mass is measured in Atomic Mass Units (amu), which are scaled relative to carbon, 12C, that is taken as a standard element with an atomic mass of 12. Each element is uniquely defined by its atomic number.Ītomic mass: The mass of an atom is primarily determined by the number of protons and neutrons in its nucleus. Boiling pointĪtomic number: The number of protons in an atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
